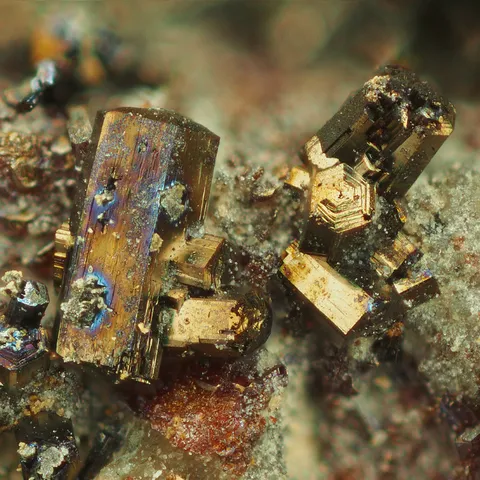ARGENTOPYRITE
Class : Sulfides and sulfosalts
Subclass : Sulfides
Crystal System : Orthorhombic
Chemistry : AgFe2S3
Rarity : Uncommon to rare
Argentopyrite is an iron and silver sulfide present in hydrothermal silver-bearing and often nickel-bearing veins. Quite rare, argentopyrite is the dimorph of sternbergite. Its name comes from the Latin argentum and pyrite in connection with its chemical composition. It occurs in small, thick, pseudohexagonal tabular prisms, frequently united in botryoidal encrustations or radiate aggregates. It is a steel-gray to pewter-white colored mineral when fresh, dulling to dark bronze, dark blue, and purple. It is an accessory ore of silver.
Main photo : Argentopyrite from Schneeberg, Saxony, Germany © Christian Rewitze
Argentopyrite in the World
Twinning and special crystallizations
Argentopyrite may exhibit pseudohexagonal and lamellar twins.
Fakes and treatments
No fake or treatments identified for this mineral species.
Hardness : 3.5 to 4
Density : 4.25
Fracture : Irregular
Trace : Gray
TP : Opaque
RI : -
Birefringence : -
Optical character : -
Pleochroism : None
Fluorescence : None
Solubility : Nitric and hydrochloric acid
Magnetism : None
Radioactivity : None